Mechanism of Action
Toxin Structure
|
Entry into the Cytoplasm
- Ricin B-chain (RTB) binds the terminal galactose residue on cell surface glycoproteins or glycolipids.
- RTB binding induces endocytosis of the entire ricin molecule.
- The ricin molecule is contained within an early endosome and transferred through the Golgi stacks to the Endoplasmic Reticulum (ER).
- At the ER, disulfide isomerase reductively cleaves the disulfide bond connecting ricin’s two subunits.
- Once separated, Ricin A-chain (RTA) becomes unstable at intracellular temperature and unfolds to mimic a misfolded protein.
- The unfolded RTA is recognized by COPII, an ER transmembrane protein, which traffics RTA back to the Golgi.
- In the Golgi, RTA is marked as a misfolded protein with ubiquitin and shuttled back to the ER to be degraded by the proteasome.
- Back in the ER, RTA binds the Hrd protein complex and is shuttled toward the proteasome.
- RTA is able to dislocate from the Hrd complex by bypassing the proteasomal cap of the Hrd1 subunit and is able to avoid degradation.
- RTA is now free in the cytoplasm and refolds into a stable, soluble, and enzymatically active structure with the aid of chaperon proteins.
Inactivation of Ribosomes
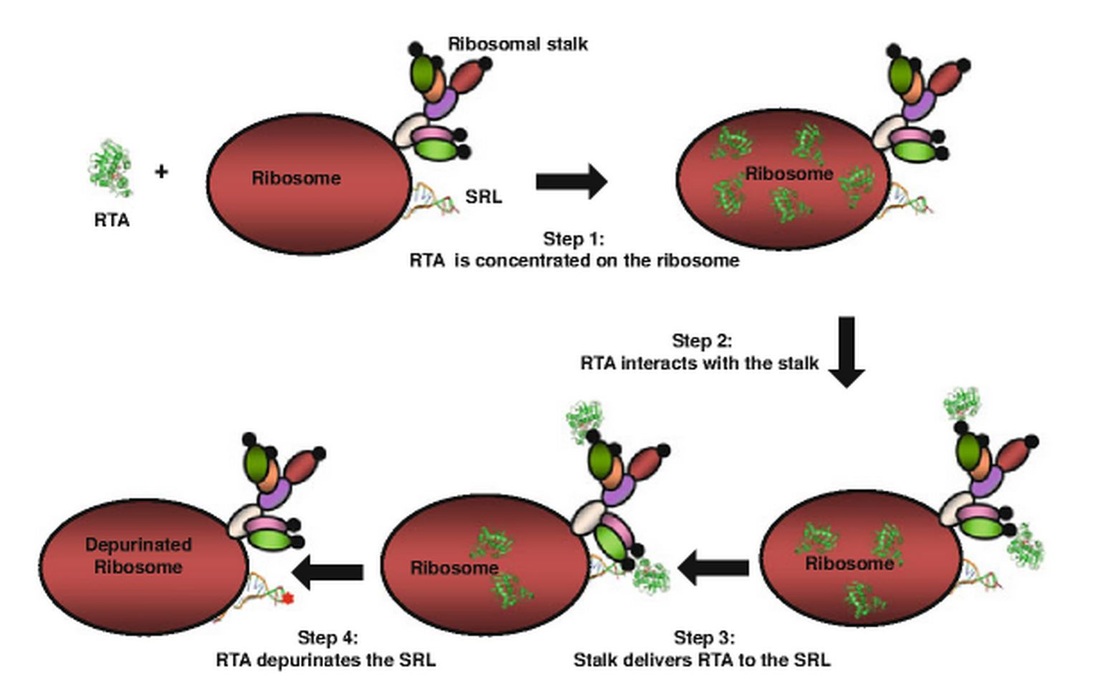
- Nonspecific electrostatic interactions draw active Ricin A-chain (RTA) molecules in the cytoplasm to the surface of the ribosome.
- Once on the ribosomal surface, secondary electrostatic interactions associate RTA with the ribosomal stalk, a distinct protein structure on the large ribosomal subunit.
- Ribosomal stalk proteins position RTA on the sarcin-ricin loop (SRL), a highly conserved stem loop within the 28S ribosomal subunit.
- RTA hydrolyzes the N-glycosidic bond between an adenosine residue and the ribose sugar at position 4,324 in the SRL.
- The depurinated SRL is unable to bind elongation factor 2 (EF2).
- Lack of EF2 binding prevents EF2-dependent GTPase activity required to drive the translocation step of protein synthesis.
- Ribosomal protein synthesis arrests and the cell dies.